Pete Farley
Director of Communications
New tool can produce colorful high-resolution videos of developing organisms that are ‘marvelous to watch’
3D rendering and video by Lachlan Whitehead/The Walter and Eliza Hall Institute of Medical Research
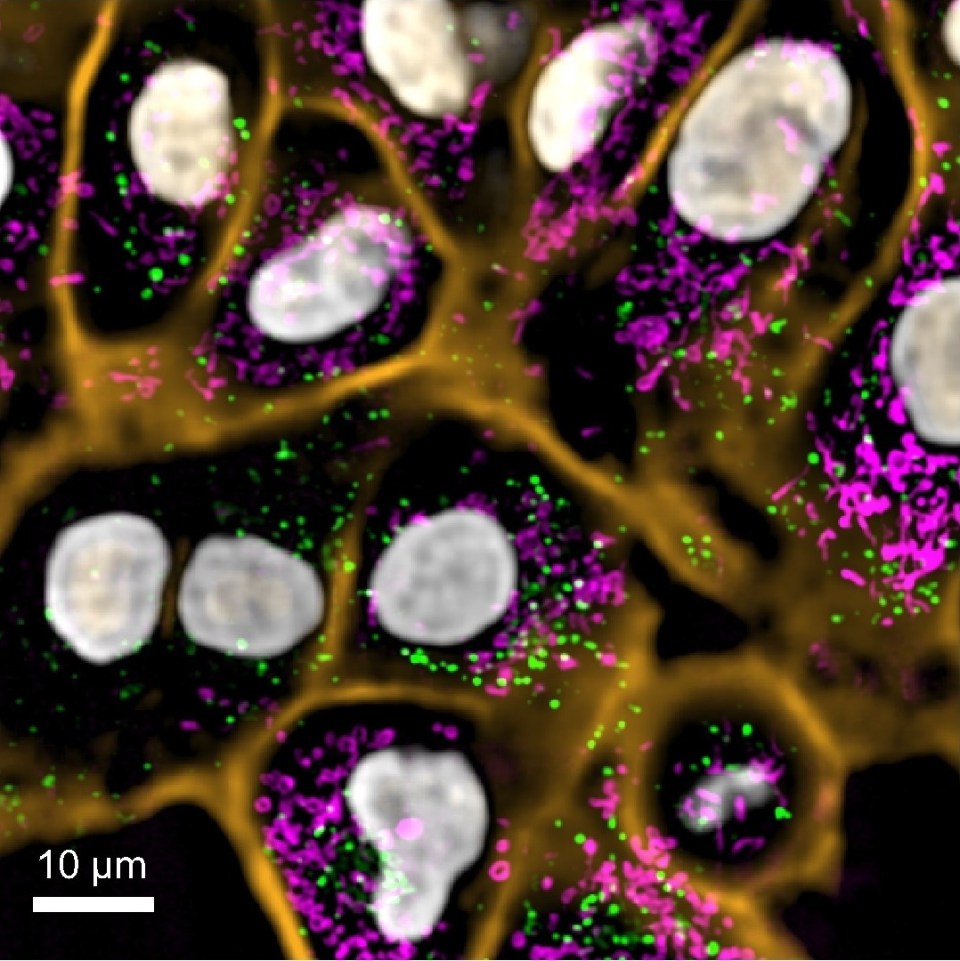
SAN FRANCISCO, CA (March 21, 2022) – When biologists use genetic tricks to make the cells of model organisms fluorescent, the scintillating images and movies that result help answer questions about ourselves—how our brain develops, how a virus might affect the early immune system, or what drugs might be useful to treat certain genetic disorders. But getting cells to shine, while an impressive feat, is just the first step in these experiments. Just as important is how scientists actually capture the light shows they’ve created, in organisms that are sometimes so small that they are just barely visible with a squint, even in later stages of development.
Now, scientists at the Chan Zuckerberg Biohub are introducing a new microscope, dubbed DaXi (pronounced “dah-shee”), that addresses long-standing challenges for researchers using model organisms such as zebrafish to study embryonic development, tissue regeneration, and more. In a Nature Methods paper published March 21, 2022, the creators report that DaXi, a so-called light-sheet microscope, can capture a space about 18 times larger than existing light-sheet microscopes, while preserving spatial and temporal resolution and solving a number of other practical problems that have plagued light-sheet microscopy in the past.
“It’s definitely an advance on the current state of the art,” says Loïc Royer, PhD, a CZ Biohub group leader in Quantitative Cell Science and senior author of the new paper. “We’ve pushed every dimension to create a practical system that has the best combined performance in terms of resolution, field of view, speed, and flexibility for biologists to design their experiments.”
Royer’s lab is one of many that work with zebrafish (Danio rerio), an important model organism whose eggs and embryos are transparent and which develops rapidly, making it easy for scientists to closely observe their organs and tissues forming in real time. An increasingly popular research tool, zebrafish are helping scientists understand why development sometimes goes awry and have even contributed to life-saving medical interventions.
Just a couple of decades ago, however, scientists’ ability to fully realize the potential of such research was substantially limited by the state of microscope technology. Traditional “confocal” microscopes used with fluorescing cells blast samples with light thousands of times brighter than the sun, which unsurprisingly damages living samples scientists would otherwise follow over time.
About 15 years ago, researchers began to turn to light-sheet microscopy, a gentler approach that sweeps a thin sheet of light across the sample, illuminating only a sliver at any given moment. The substantial reduction in light exposure afforded by light-sheet microscopes meant embryos could survive even as scientists imaged them over hours or days.
Light-sheet microscopy was a major advance for developmental biologists, but it came with problems of its own—a more complex approach means a more complex setup. Samples were now tightly surrounded by light-beam emitters and detectors, presenting a major logistical challenge: biologists simply didn’t have any room to work. With only enough space to fit a small tube containing their sample, scientists had little hope of making manipulations during their experiments, let alone scaling them up for high-throughput imaging.
Scientific inquiry frequently takes its practitioners in unexpected directions, but the practical challenges of early light-sheet microscopes left biologists with little flexibility to change their experiments once the devices, costing tens of thousands of dollars or more, were in place. For years, engineers and physicists have been working on tweaks to address this problem, but creating more room to work has always come with trade-offs including slow scanning speed, lower resolution, or a field of view too small to fit, for example, an entire developing zebrafish.
“All of the options have had drawbacks,” says first author Bin Yang, PhD, a former optical scientist in Royer’s group now at KLA, a technology firm in Milpitas, Calif. “But there’s a special need for this kind of imaging, so we weren’t satisfied just accepting the options that existed—we were really motivated to work on it.”
Named by combining two Mandarin Chinese words referring to its large imaging volume (da) and high-resolution capability (xi), the DaXi scope’s impact comes from addressing all of these problems at once, says Yang, adding that the videos one can produce with the scope are “marvelous to watch.” He detailed the following characteristics as key factors in the microscope’s success:
At least one other research group has already begun to build a DaXi microscope in their lab, and Royer and Yang hope to see it adopted by many others as well. Royer estimates that the microscope costs about half as much to build as a conventional four-objective light-sheet microscope, but he expects the cost to come down as DaXi becomes more broadly adopted and scientific and industry experts are able to lend their expertise. To accelerate this process, Royer’s group has made all the details of DaXi’s design and construction available in an open-source GitHub repository.
“With DaXi, we show that you don’t need to compromise,” Royer says. “You can run biological manipulations of a whole organism over long periods of time while getting images at amazing resolution. We think it’s a game-changer.”
Stay up-to-date on the latest news, publications, competitions, and stories from CZ Biohub.
Marketing cookies are required to access this form.
 News Releases
News Releases
Nine researchers receive unrestricted funding for projects to harness the immune system to detect disease and monitor ...
 News Releases
News Releases
The CZ Biohub San Francisco and CZ Imaging Institute will join together to develop novel imaging technologies that provide entirely new insights into human ...
 News Releases
News Releases
The growing team at CZ Biohub NY is shaping the future of life sciences in the New York ...
Stay up-to-date on the latest news, publications, competitions, and stories from CZ Biohub.
Cookies and JavaScript are required to access this form.